The Internet bunk of HHO and those who are its "analyzers"
There is nothing worse than a Journalist - who is barely a "journalist" at that - trying to write a technical article involving both Chemistry and Engine Mechanics and the fuels and fueling of them - both of which they do not understand. The result is proof of gibberish as you can find here => http://www.aardvark.co.nz/hho_fraud.shtml - written in 2008 and at least as of June 2, 2014 is still up.
We will reproduce it below in sections and tell you why it is "bunk" in each section. The title in the browser bar is "Scientific proof proving that HHO systems are a scam" and the title on the actual page is "Examining the Science behind "HHO" systems" . The inconsistency of the titles should be a give-away. No "scientific proof " is offered. We will stike through the text - unedited - so you can see what he wrote and then what we have to say. In typical Internet fashion - this erroneous article has been reproduced over and over in various forums, newsgroups, emails and on websites. Garbage in equals garbage out - and it so becomes garbage gospel.
It starts out:
It's been two months since I wrote my original articles on the subject of these "run your car on water" (RYCOW) schemes and I've been constantly researching the subject to get a better understanding of all the issues involved.
Let's see what the science says about these systems, even when we take into account the very real effect that hydrogen/oxygen enrichment can have on the efficiency of an internal combustion engine.
OK - so at least he admits he hasn't any experience or a clue about what he is about to write !!!
Next he writes:
Scientific proof
By passing an electrical current through water, you can break the bonds that hold the atoms of hydrogen and oxygen together in the form of a water molecule.
This is good science and is a process that has been known since the 1800s.
To break these bonds however, requires a significant amount of electrical energy that must come from somewhere. In the case of the RYCOW systems, this energy has to come from the vehicle's alternator and in most cases, that limits the amount of available power to around 0.75-1.0KW.
BUNK . . . where is that leap to limitation of a power analysis come from ??? So does he really mean to be saying an automotive engine only has 1.3 horsepower? 1Kw = 1000 volt-ampere = 1.341 horsepower (mechanical). REALLY ??? There are plenty of ways to to get upwards to and over 1000 amps from a properly sized and built automotive alternator, though most "stock ones" put out between 100 - to 150 amps before the diodes will avalanche.
Also, technically one does not "pass an electrical current through water", though the equation he deals with means "pure water" - de-ionized, distilled water - with no minerals in it. Technically - the current need not "pass" through the water - as distilled water - and deionized water is so resistive - it is technically considered non-conductive for lack of a better description. What REALLY happens is in the described electrolysis processes occur in pure water, H+ cations will accumulate at the anode and OH− anions will accumulate at the cathode. This can be verified by adding a pH indicator to the water: the water near the anode is acidic while the water near the cathode is basic. The negative hydroxyl ions that approach the anode mostly combine with the positive hydronium ions (H3O+) to form water. The positive hydronium ions that approach the negative cathode mostly combine with negative hydroxyl ions to form water. Relatively few hydronium (hydroxyl) ions reach the cathode (anode). This can cause a concentration overpotential at both electrodes.
Pure water is a fairly good insulator since it has a low autoionization, Kw = 1.0 x 10−14 at room temperature and thus pure water conducts current poorly, 0.055 µS·cm−1. Unless a very large potential is applied to cause an increase in the autoionization of water the electrolysis of pure water proceeds very slowly limited by the overall conductivity.
So . . . the first "boo-boo" - not understanding what electrolysis of water is, and this in itself is the gravamen to the entire article. But that is the simple mind for you - unable to get the devil in the details which then - like any math problem causes all the rest of the analysis to be faulty. We have seen many many website locations repeat this misnomer - that an alternator cannot produce enough power to make enough gas and power the car too. BUNK.
Then he writes:
Of course that 1KW or so also has to recharge the vehicle's battery, power the ignition, headlights etc, and run the various other accessories that abound in a modern car or truck.
So it's safer to assume that only around 600W is always available for the electrolysis cell, which is a maximum of around 50 amps.
Now if we delve into some more good science we find this formula for Faraday's First Law:
Vtheoretical (in m3) = (R I T t) / (F p z)
Where R=8.3414 Joule/(mol Kelvin), I=current(in amps), T=temp in Kelvin, t=time (in seconds) F=Faraday's constant = 96485 Coulombs per mol, p=ambient pressure (pascals), z=num of excess electrons.
If we plug some numbers into this formula and do some simple conversions then the following results appear:
237.1KJ is required to convert 1 Mole of water (18g) to H2/O2To convert 1 Mole of water to gas via electrolysis will reuire 237.1KJ of energy1 litre of H2O produces 55.55 Moles of H2 (1,358.3l) and 27.775 Moles (679.15l) of O2 (for a total volume of 2037.45l of H2/O2)To convert 1 litre of H2O to H2 and O2 by electrolysis will require 3.658KWH
Of course all the above assumes a 100% efficiency and as we know this is simply not attainable with current materials and technology.
WOW . . . pure gibberish. All based on that erroneously made statement of that solely available 1Kw of power available. Uh . . . 600 W - as he uses it - is more like 6 - 10 amps as he writes it - but let us use his "50 amps" figure (@ 12 volts DC) he also assumes the "1 liter" of water is converted instantaneously - and does not account for the time over which the electrolysis occurs. He "assumes" these standard pure water calculations - which in reality are greatly affected by the fact no one uses laboratory grade pure water - and rather "conductive forms" of water, and water based fuel stocks for a Hydrogen Generator from pure water to salt water - and its salt derivatives - and the alcohols - all produce from 50 to 400% more hydrogen at often 50 to 100% LESS energy depending on the build and design of the Hydrogen Generator.
In an Engineering Physics class he would get an "F-" - for so bad and worse - as to an analysis even without the math errors.
Next he writes - and it just gets worse from here . . .
To get an idea as to how efficient the electrolysis process is when performed in a home-made electrolyser I found plenty of videos on YouTube where people proudly share their results.
This example is pretty typical so let's do the math:
12V x 40A ==> 480W
If we plug Faraday's First law into this figure, it suggest that in a 100% efficient cell we should be generating around 4.5 liters per minute of H2/O2 from 480W of electrical energy.
The cell in the YouTube video produces just 2.0 liters per minute, indicating an actual efficiency of around 44% - so we'll assume 50% efficiency for the rest of our calculations.
Now we need to take a look at some of the scientific research that demonstrates how hydrogen/oxygen enrichment can improve engine efficiency. This report (PDF) suggests a best-case improvement in thermal efficiency of around 14.8%-15% so we'll use the higher figure.
Now let's plug in some other figures and see if we can get a net-gain.
The engine in that paper was producing around 6.5HP at 1,500RPMs and needed 240 liters per hour (4 liters per minute) of H2/O2 gas.
An average auto engine will be capable of around 200HP and so a simple scaling would indicate that 30 times the 4lpm figure (or 120lpm) would be required to see the same 15% increase at full throttle. Of course we don't drive around at full throttle all the time so a safer figure to use would be the 20HP or so that an average-sized car requires to cruise at highway speeds.
20HP / 6.5HP = 3 times the horsepower so we'll need 3 times the fuel and three times the H2/O2 gas to see the same effect. That brings our H2/O2 requirement to 12.0 lpm.
Actually, at this stage we must also take into consideration that the engine in the paper was as diesel which uses significantly less fuel to create the same HP (due to the higher compression ratios and naturally more efficient cycle) -- that's why diesel vehicles get better MPGs than gasoline-powered ones. So we'll allow a very conservative additional 20% to account for the greater mass of gasoline required to produce the same power.
That lifts the H2/O2 gas requirement to 14.4 lpm.
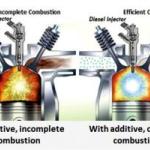
He then now cites "YouTube" - really - the junkyard of information of the Internet where all the "yahoos" report their findings - and that first thing comes to haunt him - no one is using "pure water" and no one is producing "pure hydrogen". He has lost all scientific credibility at this point and - of course he does not know this or he would not continue. He has mixed Apples and Oranges . "HHO" gas is not Hydrogen - it is Oxy-Hydrogen gas.
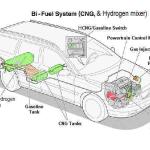
One cannot fuel a vehicle at the required stoichiometric mixture needed with a PRE-MIXED gas. Also he fails to take into account that a lawnmower engine might run on it - but an automobile has a complex emission control - almost certainly fuel injected and Oxygen Sensor controlled system that can ONLY be bi-fueled and that changes everything in his "proof" to gibberish.
Can an engine run on Hydrogen ??? Absolutely and it is only lawful for the engine manufacturer to create a fueling system to do so - and Ford has a production supercharged V-10 Triton PURE Hydrogen engine it puts in its cut away bus chassis for use as an Ultra Low Emission Vehicle (ULEV) for use around airports and the like.
Well for the final nail in his own coffin he boldly writes:
We're assuming a 20HP requirement for cruise speed so once we add the 14.4 lpm of H2/O2 the engine will output an additional 20 x 0.15 = three horsepower.
So far so good.
Next we have to work out how much energy we'll be taking back in order to create those 14.4lpm of H2/O2.
If we use Faraday's First Law again to calculate that figure we discover that we need around 1,536 watts - but that's only if our electrolysis cell was 100% efficient, which it's not.
We've assumed an efficiency (based on real-world figures) of just 50% for the electrolysis process so we need to double that figure -- which gives us 3,072W of electrical energy required to generate sufficient H2/O2 to give us the maximum fuel-efficiency improvement at cruise speeds.
That 3,072W is 4.1 horsepower.
Uh-oh, it looks like we're spending 4.1 horsepower creating H2/O2 gas but only getting 3 horsepower back. That's a net *loss* of 1.1hp which mean's we'll actually burn *more* fuel than if we hadn't gone to all this trouble.
But wait... it gets worse...
That 4.1hp assumes that our alternator is 100% efficient in turning mechanical energy into electrical energy -- which it certainly is not.
An auto-style alternator is actually only around 60% efficient so the actual amount of horsepower we'll be sucking from the engine to power our cell is going to be around 6.8hp.
Now we find that the net energy return from these "HHO" systems is an even bigger. We're spending 6.8 horsepower to boost the engine's output by just 3 horsepower.
That missing 3.8 horsepower will have to come from burning *MORE* not less gasoline.
I'm sorry -- but those are the facts, there is the science.
I invite anyone to pick holes in my science or my math.
Well - let's see - math doesn't run a vehicle or engine; and engineers will tell what is on paper is not the way it really works; but in this case none of this has made sense from the science point of view as he has mixed everything up. A "net loss" of horsepower - REALLY?? One could argue - and rightly should argue the costs in energy of oil exploration, refining and transportation is all a "net loss" and it is - but at east that makes sense and not conjecture based on a premise he had wrong in the first place.
Anytime a diesel uses less fuel to run, you are saving THAT fuel - but the real question is what is the fuel UNDER LOAD that replaced it doing to create the horsepower - plenty of REAL studies have been done showing how Methane and Hydrogen work in a Diesel engine and even how Hydrogen alone works - but let us face it -the main flaw is "HHO" - as no one in their right mind would waste time measuring in a study the effects of an engine output using a pre-mixed fuel - as "HHO" is.
Hydrogen works as "pure" hydrogen - and mixed with CNG to create Hythane - a.k.a. "HCNG" in varying amounts from 10% to 90%, and his article does not even identify what it is dealing with as a fuel or any other effects beneficial or non - on the engine operation. It is pure Internet "BUNK">
We accepted his invitation to pick holes in his math - and it turns out gaping ones were ALREADY there with his general nonsense in the first place trying to write about something he admits he does not understand as to engine mechanics and emissions systems. Yes "HHO" does not work well enough to be worth the investment and trouble to install it - but just not for the reasons this guy has mixed up and made up.

 ,
,  ,
, 

 ,
,  ,
, 

 ,
,  ,
,  ,
, 
 ,
,  ,
, 

 ,
, 




 ,
,  ,
,  ,
,  ,
,  ,
, 
 ,
,  ,
,  ,
, 



 ,
,  ,
,  ,
,  ,
, 






 ,
,  ,
,  ,
,  ,
,  ,
, 











 ,
,  ,
,  ,
, 




 ,
, 

 ,
, 





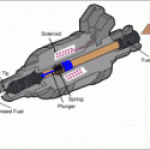



 ,
,  ,
,  ,
,  ,
, 



 ,
,  ,
, 






 ,
,  ,
,  ,
,  ,
, 

 ,
,