ANSWERS: NOx and the diesel
NOx emissions – formation, reduction and abatement
The two most significant pollutants produced by humans (anthropogenic) are NOx emissions and particulates. Information on particulates can be found here.
This page original credit => http://clean-carbonenergy.com/nox-emissions.html
Q. What does the term NOx mean?
A. It refers to nitrogen oxides. The purists would say that it refers to nitric oxide (NO) and nitrogen dioxide (NO2) only, but most also include nitrous oxide (N2O) in this description. There are some other variants, but their concentrations in the atmosphere are too low.
Q. Why are NOx gases produced?
A. There are three main causes of NOx emissions:-
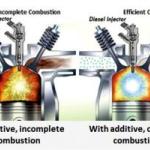
- High temperature combustion of fuels where the temperature is hot enough (above about 1300°C/ 2370°F) to oxidize some of the nitrogen in air to NOx gases. This includes burning hydrogen, as it burns at a very high temperature. Comments on diesel engines are shown below.
- Burning plant material releases nitrogen oxides, as all plants contain nitrogen.
- Chemical and industrial processes which use nitric acid, nitrates or nitrites will release NOx gases.
Q. What is the difference in combustion between a diesel engine and a petrol/gasoline engine?
A. In a petrol/gasoline engine, a mix of fuel and air is drawn into the chamber. This is compressed and then ignited by a spark plug. (Unless it is a direct injection gasoline engine)
In a diesel engine air is drawn into the cylinder, and is compressed by around twice as much as in a petrol/gasoline engine. This compression generates heat, so that diesel fuel burns spontaneously when it is injected.
Q. Why do diesel engines produce more NOx than petrol engines?
A. Diesel engines operate at a higher temperature and pressure than petrol engines. These conditions favor the production of NOx gases. The quantity depends on the volume and duration of the hottest part of the flame. In the United States the "answer" to this has been to blend lower temperature burning diesel fuels than previous refinements of Diesel. Also Hydrodesulfurization - the process used in making ULSD takes out some BTU power. there is also an effort to make diesel from plant stock and animal parts rendering left over from the slaughter process for our food which has higher cetane - but also lower BTU per gallon.
Q. Why are diesel engines more fuel efficient than petrol/gasoline engines?
A. Diesel fuel produces more energy for a given volume (diesel has a lower calorific value, but a higher density than petrol/gasoline). Also the higher combustion temperature in a diesel engine makes it more efficient. Heat engines can generate more useful work if they operate at higher temperatures.
Diesel and gasoline have roughly the same energy per unit mass (lower heating value), about 41 MJ/kg41 MJ/kg).
The density of diesel is about 833 kg/m3833 kg/m3 compared to 740 kg/m3740 kg/m3 for gasoline. This gives diesel about 13% higher energy density per volume. BUT the real issue is how to release that energy - and spark ignition does not work because diesel fuel does not "atomize" or "vaporize" like gasoline does and that means the vapors will not ignite from Diesel fuel. Both Gasoline and Diesel however will spontaneously ignite - a.k.a. "explode" if injected into an environment that is under pressure and hot/ this means a Diesel engine will also run on Gasoline - HOWEVER - the lubrication of the Diesel fuel serves a function to lubricate the injection pump system that injects it - whereas Gasoline as a "solvent" more than the oily - slippery nature of traditional Diesel meant it would heat up the injection pump and destroy it due to friction.
Q. How do you reduce NOx emissions from diesel engines?
A. By lowering the combustion temperature, typically by Exhaust Gas Recirculating (EGR). Some exhaust gas is cooled and injected back into the combustion chamber. There is less oxygen in the exhaust gas because some has been consumed by previous combustion, so there is not as much to feed the flame. The exhaust gas also has a higher heat capacity than air, so it takes longer to heat up. The easiest and simplest way to reduce NOx is to cause the combustion to occur in a humid environment/ Most would call it "water injection" or "vapor Injection"; and what happens is in the presence of H2O - or a H2O based or soluble liquid pyrolysis occurs which breaks the H2O bond that then as H2 and O2 in the gaseous form that instantaneously burns in a cooling reaction that tends to prevent NOx formation but "can" yield actually MORE energy. Google search "diesel water injection system" to learn more. read with a grain of salt because more people selling kits and talking about this - do not know what they are talking about. HOWEVER straight water injection - that is not in the form of a fuel like alcohol - WILL cause ignition delay with poorer lower cetane diesel fuel, while still reducing NOx formation; however the engine will loose power. The trick is to super heat the H2O based or soluble liquid vapor to the near pyrolysis point so that upon injection of the Diesel all fuel in the cylinder ignites and explodes to burn immediately. This is why Oxyhydrogen which is a mixture of hydrogen (H2) and oxygen (O2) gases.has had good results in reducing NOx formation - it is commonly called "HHO". However if not metered properly to an engine - it will in greater amounts actually increase NOx formation.
Anyone can research metanol injection and discover both methanol and ethanol - both alcohols can be used as vapor injection. Methanol is also recognized as wood alcohol, methyl alcohol, and methyl hydrate. It is the common alcohol used in "alcohol-burning" race cars. It is commonly used as anti-freeze in -20 degree f blue winter blend windshield washer fluid as well as a gas line dryer in Heet and for keeping air lines dry in trucks with airbrakes. If used as the sole fuel in a diesel, it requires an ignition source as it won't auto-ignite since its auto-ignition temperature is close to 900 degrees f. Thus it could also be used as pre-turbo fuel metered injection system tied to the accelerator pedal to bi-fuel an engine - both gasoline or diesel.
Q. Are there any other consequences of using EGR?
A. Yes, there is a downside. As the combustion temperature drops, so does the power, and the fuel economy.
Q. How can you remove NOx from exhaust gases?
A. There are various techniques, depending on the applications, although a lot of effort goes into designing burners which reduce NOx emissions in the first place.
- Selective Catalytic Reduction (SCR) is the most common method in diesel vehicle exhausts, but it is expensive so isn’t use in small cheap vehicles. There are various proprietary blends of ammonia and urea which can be injected into the exhaust flow. These react with NOx gases over a catalyst, which turns them into harmless nitrogen and water.
- Selective Non-Catalytic Reduction (SNCR) – takes place in ducting where the temperature is about 1000°C (1800°F). Urea or ammonia is injected, and the NOx gases are reduced to nitrogen without the need for a catalyst. This is in use on many large diesel engines - by law in the US - and they use what is called DEF - Diesel Exhaust Fluid which is about 30% urea Diesel exhaust fluid (DEF) is an aqueous urea solution made with 32.5% urea and 67.5% deionized water.
- On an industrial scale, exhaust gases can be scrubbed with chemicals such as sodium hydroxide, hydrogen peroxide, or a mixture of hydrogen peroxide and nitric acid. These chemicals react with the NOx gases and removes them.
Q. Why are NOx gases harmful?
A. Internal combustion engines can produce all three nitrogen oxides.
Nitrous oxide (N2O), also known as 'laughing gas'.
- It is a serious greenhouse gas, and is defined as being 298 times as bad as CO2 because of its radiative effect, and the time taken to break it down.
- Used as an anaesthetic and generally considered to be non-toxic. It does react with vitamin B12, which may be a problem for those who are deficient.
- It is broken down in the stratosphere, and catalyses the breakdown of ozone. Ozone in the upper atmosphere is vital for absorbing UV rays; at the earth’s surface, it is harmful.
Nitric oxide (NO).
- Readily oxidised in the atmosphere to nitrogen dioxide.
- Non-toxic in small quantities, infact it serves a vital role as a regulator within the human body.
Nitrogen dioxide (NO2).
- A major pollutant and component of smog. Its brown fumes may be familiar from school chemistry experiments.
- It reacts with water to produce nitric acid, which is why it is so irritating to the eyes and respiratory tract.
Q. What are SOx emissions?
A. When fuel is burnt in an engine, any sulphur will be converted into sulphur dioxide (SO2) gas. This readily dissolves in water to produce an acid, which accounts for the irritation to your respiratory tract if you inhale it. It also affects the ecology. Oil and gas in the ground can contain large quantities of sulphur, which have to be removed in the refinery. Some countries have lax regulations on sulphur content in fuel, with resulting high pollution levels.
Q. What is smog?
A. Fog or haze intensified by smoke or other pollutants.
- Traditional smog was caused by burning coal, particularly high-sulphur coal.
- Photochemical smog. Nitrogen dioxide, and a fellow pollutant volatile organic compounds (VOCs), combine in the presence of sunlight to produce ozone and a variety of other compounds. These have a nasty affect on the respiratory system. More detail on photochemical smog.




 ,
,  ,
, 

 ,
,  ,
, 

 ,
,  ,
,  ,
,  ,
,  ,
, 
 ,
, 

 ,
,  ,
,  ,
,  ,
, 




 ,
,  ,
,  ,
,  ,
, 

 ,
,  ,
,  ,
,  ,
, 













 ,
,  ,
,  ,
, 




 ,
,  ,
, 

 ,
, 
 ,
,  ,
,  ,
,  ,
,  ,
, 

 ,
,  ,
,  ,
, 




 ,
, 







 ,
,  ,
,  ,
,  ,
, 




 ,
, 

 ,
,  ,
,  ,
, 






